This post is also available in Dutch.
Scientists have been working on the development of cell replacement therapies for decades. Are we finally close to a break-through?
Cell replacement therapy (CRT) might be one of the most promising future therapies for a wide range of conditions that develop due to the dysfunction or death of a certain type of cell in the body. One example of such an illness is Parkinson’s disease. Motor symptoms often present in Parkinson’s disease, such as shaking, stiffness and slowness of movement, are caused by the death of a particular type of cell in the midbrain. These cells normally produce dopamine, a substance which plays a role in signal transmission in the brain. As a result of cell death, there is a deficit of dopamine in the brain, which strongly undermines the brain’s ability to properly control movement. However, in the future scientists might be able to restore these cells, and help patients get back on their feet, using CRT. Multiple research institutes are currently setting up clinical trials to investigate if this treatment could relieve Parkinson’s symptoms so it is worth taking a closer look at this therapy and its potential.
What is cell replacement therapy and how does it work?
The idea of cell replacement therapy (also called stem cell therapy) sounds quite simple. We know that cell death in the midbrain causes motor impairments in Parkinson’s disease so why don’t we just try to replace those cells? That means placing new healthy dopamine-producing cells in the part of the brain where the dead neurons used to release dopamine. As research in monkeys with a form of Parkinson’s disease has shown, transplanted cells can functionally integrate themselves into the brain. In addition, monkeys who received the transplant showed fewer Parkinson’s symptoms and more spontaneous movement up to two years after the transplantation. During this period, implanted cells survived in the monkey’s brain and did not produce any dangerous effects in the body.
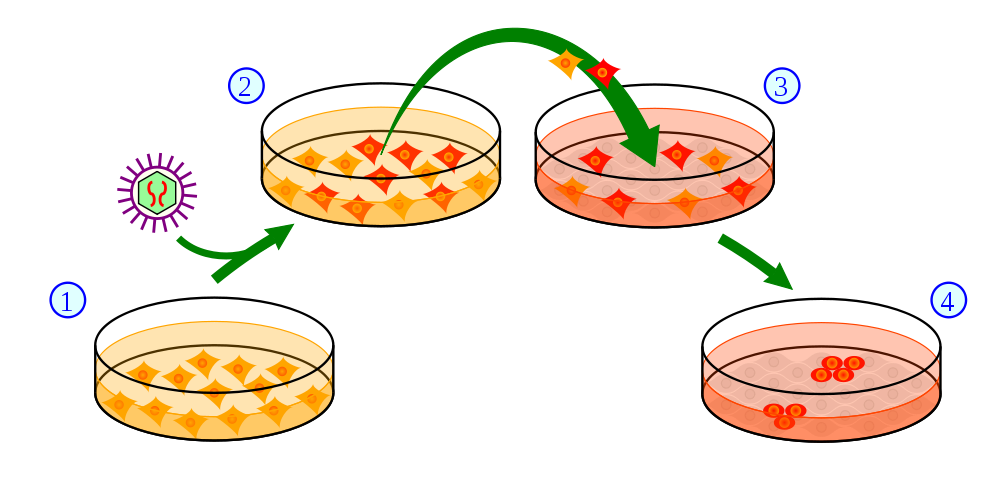
Although there are multiple techniques available to create these new brain cells, we will focus on one particular approach here to generate dopamine-producing brain cells, while the brain itself is not capable of producing them anymore. This is where a special development called “induced pluripotent stem (iPS) cells” comes in. A word by word analysis of this term can already give us some idea of what an iPS cell is. First, stem cells are cells that can still specialize and turn into different types of cells (e.g., a liver cell, brain cell). The word pluripotent (pluri = many, potent = powerful, effective) indicates that the stem cell can give rise to almost any of the cell types that make up the body. These properties can be artificially induced by scientists in cells growing in a dish. In most cases scientists use adult skin cells from the patient themself and convert them into iPS cells. This is done by exposing the isolated skin cells to certain chemicals, which stimulate the cells in such a way that they revert to their original immature unspecialized state, thereby becoming an iPS cell. Once in this state, scientists can transform those stem cells into various types of cells (requiring very intricate knowledge of the specific environments that will produce these cell types). This way they can create a patient-specific transplant of dopamine-producing neurons.
Why do researchers think that this might be a promising treatment for Parkinson’s disease?
Current treatments for Parkinson’s disease address the symptoms, but not the underlying cause of the disease. CRT, however, does aim at fixing the underlying biological problem. Therefore, it represents one of three crucial components that are needed to cure Parkinson’s disease, which are: a disease-halting mechanism, something that prevents further damage to brain cells, as well as some form of CRT. Moreover, many current treatments just temporarily suppress the symptoms, while CRT might be able to counteract disease progression, bringing dopamine production back to higher levels and improving movement. Another advantage is that there is little risk of an immune rejection of the transplant, as the cells are coming from the patient themself. Therefore, there is no need for immune-suppressors.
Why has it taken decades of research to develop these therapies and are they safe for humans?
It is important to stress that cell transplantation for Parkinson’s disease (and other illnesses) is still experimental. Although the latest animal research suggests that the procedure is safe and can move on to human trials, we can’t be certain that the therapy will have no dangerous side-effects. The most serious concern regarding CRT, especially in the brain, is that transplanted cells might develop into tumors. Apart from this serious risk, we have not yet reached the point where CRT is feasible: Creating iPS cells is still a very expensive and technically challenging procedure. Generating iPS cells, tailor-made for individual patients, can take months, so it is not a particularly fast process and could not be applied to a larger population of patients. Nonetheless, there are currently three clinical trials conducted for cell transplantation in Parkinson’s disease (for more information click here). It is also unsure how well current CRT procedures will work in Parkinson’ patients, for example: how well these cells can integrate themselves into the existing network and if all functions can be properly restored. Still, putting all these challenges that scientists have to overcome in the future aside, there is hope that someday this technique will improve the lives of many patients.
Written by Eva, edited by Monica
Image by Y tambe – Y_tambe’s file, CC BY-SA 3.0, https://commons.wikimedia.org/w/index.php?curid=3210604